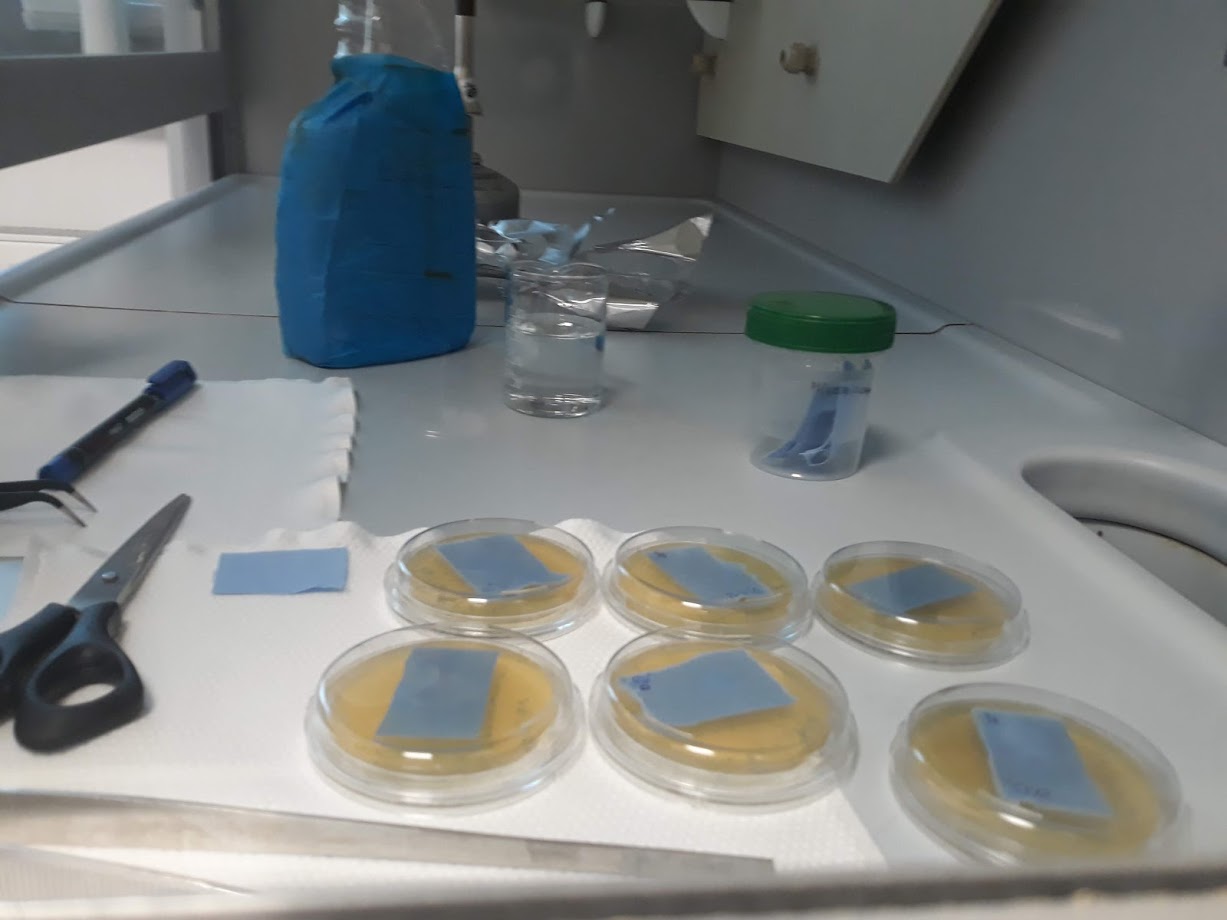
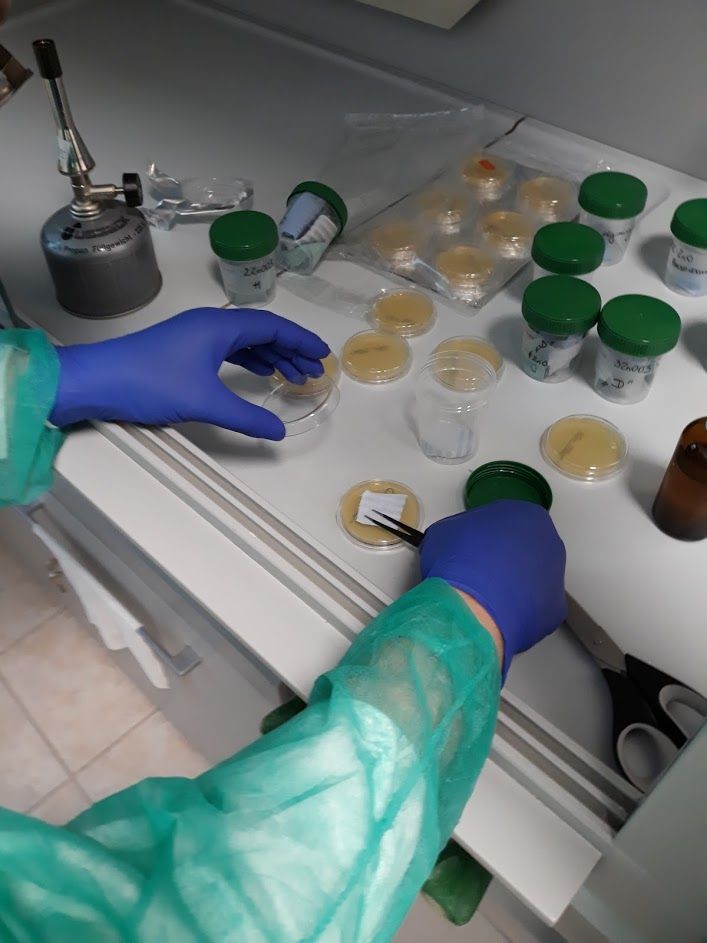
Nowadays more and more attention is put on microbiological purity of materials. Microbiological monitoring enables reduction of biological hazards.
In InPhoCat, we perform qualitative and comparative microbiological tests of non-sterile materials in aerobic conditions, consisting of:
-
-
- control and assessment of microbiological cleanliness of the surface,
- control and assessment of microbiological purity of water,
- control and assessment of microbiological purity of environmental samples.
-
The tests are intended to evaluate the efficiency of antimicrobial protection offered by catalytic and photocatalytic products. We perform also tests tailored to the customers needs. Currently, we are able to conduct research on:
-
- on ready-made agar media in Petri dishes by colony counting (related to ISO 27447: 2009)
- bioluminescence method using a certified analyzer, where functional tests are carried out on surfaces and in solutions using certified swabs (e.g. LuciPac Pen, LuciPac Pen-Aqua).
The detailed methodology is discussed and adjusted depending on the nature of the sample and the clients’ needss.